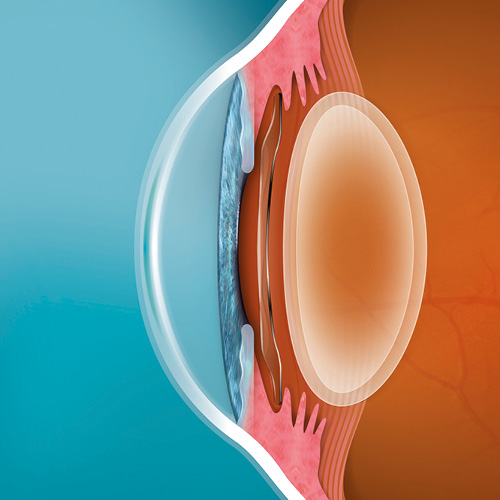
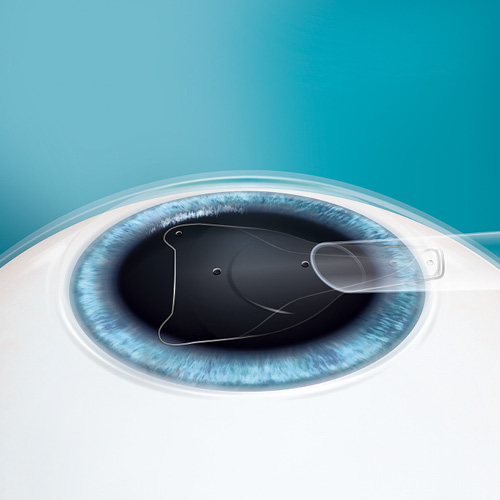
EVO ICL
STAAR SurgicalExciting new chapter in refractive surgery
EVO offers patients an exciting new chapter in refractive surgery:
- Does not induce dry eye syndrome4
- Rapid visual recovery3,6
- Superb quality of vision1
- Excellent night vision5
- No risk of ectasia6
- Improved quality of life5
STAAR’s exclusive, premium Collamer® material
EVO uses STAAR’s exclusive, premium Collamer® material
- Collamer is derived from two words that describe the composition of the material: “Collagen” and “Copolymer”.
- Collamer is a copolymer of poly-HEMA and collagen, that offers UV protection.
- Collamer has a proven history of over 20 years with more than 2 million ICL lenses distributed worldwide.
- Collamer minimizes inflammation, flare and cellular reaction.8,9
Why is the EVO ICL family of lenses* (EVO) a Game Changer?
- High rate of patient satisfaction1
- Preserves the cornea and crystalline lens3
- Low rate of adverse events1
- Outstanding safety and effectiveness1,2
- No preoperative peripheral iridotomies (PIs)
- Outstanding postoperative uncorrected visual acuity1
* The EVO family of lenses includes EVO ICL, EVO Toric ICL, EVO+ ICL and EVO+ Toric ICL.
Great for you, great for your patients
Effectiveness
The effectiveness of EVO is demonstrated by the high levels of postoperative uncorrected visual acuity, refractive predictability and stability1 enjoyed by patients. Efficacy Index is defined as UCVA (Uncorrected Visual Acuity) after treatment divided by CDVA (Corrected Distance Visual Acuity) before treatment (UCVA post/CDVA pre).
Refractive Stability
Excellent refractive stability.
IOP Stability
Intraocular pressure (IOP) is well controlled.
Predictability
High predictability across a large diopter range.
Rotational Stability
Of 629 eyes implanted with EVO, only one eye with a toric lens required surgical repositioning for residual astigmatism (Data from U.S. FDA Clinical Trial).7
Excellent Night Vision
EVO provides postoperative improvement in mesopic contrast sensitivity (CS) with and without glare.5
Low Higher-Order Aberrations
Low induction of coma-like, spherical-like and total higher-order aberrations (HOA).10
Safety
Safety data suggest reduced rates of anterior subcapsular cataract and pupillary block relative to earlier models.1,7
REFERENCES
- Packer M. The Implantable Collamer Lens with a central port: review of the literature. Clinical ophthalmology (Auckland, NZ). 2018;12:2427-38.
- Kamiya K, Shimizu K, Igarashi A, Kitazawa Y, Kojima T, Nakamura T, et al. Posterior chamber phakic intraocular lens implantation: comparative, multicentre study in 351 eyes with low-to-moderate or high myopia. Br J Ophtalmol. 2018;102(2):177-81.
- Kohnen T. Phakic intraocular lenses: Where are we now? J Cataract Refract Surg. 2018;44(2):121-3.
- Ganesh S, Brar S, Pawar A. Matched population comparison of visual outcomes and patient satisfaction between 3 modalities for the correction of low to moderate myopic astigmatism. Clin Ophthalmol. (Auckland, NZ). 2017;11:1253-63.
- Martínez-Plaza E, López-Miguel A, López-de la Rosa A, et al. Effect of the evo+ visian phakic implantable collamer lens on visual performance and quality of vision and life. Am J Ophthalmol. 2021;226:117-25.
- Wei R, Li M, Zhang H, Aruma A, Miao H, Wang X, et al. Comparison of objective and subjective visual quality early after implantable collamer lens V4c (ICL V4c) and small incision lenticule extraction (SMILE) for high myopia correction. Acta Ophthalmol. 2020;98(8):e943-e50.
- Packer M. Evaluation of the EVO/EVO+ Sphere and Toric Visian ICL: Six month results from the United States Food and Drug Administration clinical trial. Clinical Ophthalmology. 2022;16:1541-53.
- Schild G, Amon M, Abela-Formanek C, Schauersberger J, Bartl G, Kruger A. Uveal and capsular biocompatibility of a single-piece, sharp-edged hydrophilic acrylic intraocular lens with collagen (Collamer®): 1-year results. J Cataract Refract Surg 2004;30(6):1254-8.
- Brown DC, Ziemba SL. Collamer® intraocular lens: clinical results from the US FDA core study. J Cataract Refract Surg. 2001;27(6):833-40.
- Igarashi A. Posterior chamber phakic iols vs. Lasik: Benefits and complications. Expert Review of Ophthalmology. 2019;14(1):43-52.
More information
Simovision BV
Chasing Innovation in Ophthalmology
Discover the range
Contact us
Vriendschapsstraat 30
3090 Overijse
Tel.: +32 (0)2 769 70 00
VAT: BE 0875.942.068
IBAN: BE45 0014 6618 3389
FAMHP: BE/CA01/1-05918
SRN: BE-IM-000003338
A Hilco Vision Company | Simovision is a registered Trademark | Sales Conditions | Disclaimer & Privacy | Developed by Capptain